A research team led by the United States Army Medical Research Institute of Infectious Diseases, or USAMRIID, that includes Scott Pegan at the UC Riverside School of Medicine has discovered an important protective antibody target against Crimean-Congo Hemorrhagic Fever virus, or CCHFV. Their work, which appeared online yesterday in the journal Nature Communications, could lead to the development of protective antibodies for infected patients.
“Exposure of hospital workers and overseas service personnel to CCHFV is a major problem, therefore an antibody-based drug that protects against exposure from existing strains of CCHFV, while also providing therapeutic protection is ideal,” said Pegan, a professor of biomedical sciences.
CCHF is considered a priority pathogen by the World Health Organization, or WHO, as it is an emerging zoonotic disease with a propensity to spread. It is also endemic in large portions of the world. CCHF outbreaks have a mortality rate of up to 40 percent. Originally described in Crimea in 1944–1945, and decades later in the Congo, the virus has recently spread to Western Europe through ticks carried by migratory birds. The disease is already endemic in Africa, the Balkans, the Middle East, and some Asian countries. CCHFV is designated as a biosafety level 4 pathogen (the highest level of biocontainment) and is a Category A bioterrorism/biological warfare agent. There is no vaccine to help prevent infection and therapeutics are lacking.
USAMRIID has worked with CCHFV going back several decades. During this work, the institute amassed an extensive collection of mouse monoclonal antibodies against CCHFV. However, suitable animal model systems to study antibody protection against CCHFV were only recently developed, some at USAMRIID. In previous work, USAMRIID scientists Aura R. Garrison and Joseph W. Golden studied these collections of monoclonal antibodies in newly developed animal systems to identify a novel CCHFV therapeutic antibody target called glycoprotein 38, or GP38. GP38 was previously identified by the U.S. Centers for Disease Control and Prevention, or CDC, as a viral protein with unknown function.
In the current work, Garrison and Golden explored the protective efficacy of a CCHFV monoclonal antibody targeting the nucleocapsid protein, or NP. The scientists found that the NP targeting antibody protected mice against an otherwise lethal infection. This included providing substantial protection against a strain of CCHFV taken from a lethal human case. This work, therefore, opens another new direction for immunotherapeutics targeting CCHFV. The group is currently exploring the mechanism of protection of anti-NP antibodies.
“We hope to combine antibodies targeting NP with those targeting GP38 to produce a robust immunotherapeutic cocktail that can protect humans,” Garrison said.
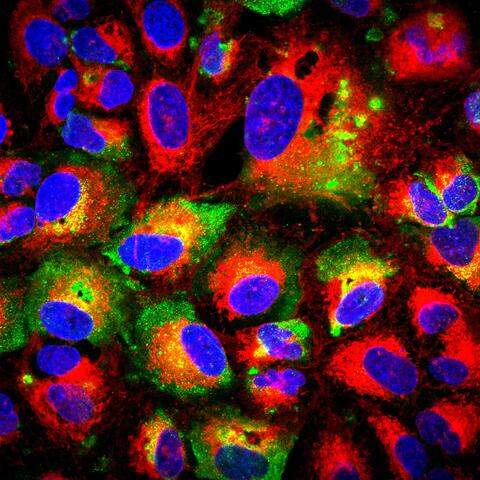
The antibody used for these studies is called MAb-9D5 and prior to this study was used as a laboratory reagent. NP is generally not thought of as a key target for protective antibodies because it is buried within the virus structure and not believed to be expressed in a manner accessible to protective antibodies. The work in this study revealed that NP is present on the surface of infected cells, which may explain why the antibody is able to provide protection. Pegan and Eric Bergeron of the CDC showed that NP-targeting antibodies can interact with NP from several CCHFV strains suggesting that this product could protect against most strains of CCHFV circulating throughout the world.
The research was funded by grants to Golden and Garrison from the Military Infectious Disease Program. Pegan and Bergeron were funded with grants from the National Institutes of Health and the Department of Defense.
Pegan, Garrison, Golden, and Bergeron were joined in the study by Vanessa Moresco, Xiankun Zeng, Curtis Cline, Michael Ward, Keersten Ricks, Scott Olschner, Lisa Cazares, Elif Karaaslan, and Collin Fitzpatrick.
This release is a slightly modified version of a USAMRIID news release.
Header image shows surface expressed nucleocapsid protein on infected cells. (USAMRIID/Kevin Zeng).