Fuels made from agricultural or forestry wastes known as lignocellulosic biomass have long been a champion in the quest to reduce use of fossil fuels. But plant cell walls have some innate defenses that make the process to break them down more difficult and costly than it could be.
In a leap forward that could be a game changer for understanding how plant biomass can be more efficiently broken down, a research team at the University of California, Riverside have joined forces with teams at Oak Ridge National Laboratory and the University of Central Florida to create a chemical roadmap to breach these defenses.
In order to access the energy-rich sugars found in the plant cell walls, researchers have renewed focus on solvating lignin, a complex polymer also found in plant cell walls that acts as a natural shield, blocking both chemical and biological attack. Lignin is particularly effective in preventing commercial enzymes from digesting cellulose, which makes up the bulk of sugars found in biomass.
In the past, different specialized chemicals and pretreatment methods have been used to improve enzyme access to cellulose but were ineffective at removing lignin. The use of strong acids, ionic liquids, ammonia, and sulfite treatments have somewhat improved the digestibility of cellulose, but these methods also leave lignin behind, making cellulose expensive to recover. Other methods have applied co-solvents such as ethanol and acetone solvate to remove lignin, but they require very high reaction temperatures that also cause the remaining sugars to degrade.
As a result, economically viable methods of transforming biomass into biofuels have yet to be realized.
Charles Cai, an assistant research engineer at the Center for Environmental Research and Technology in the Marlan and Rosemary Bourns College of Engineering at UC Riverside, and Abhishek S. Patri, a doctoral student in chemical and environmental engineering, led a team of researchers taking a new direction to focus on identifying highly specialized co-solvents, substances added to a primary solvent to make it more effective, that can facilitate milder temperature solvation and release of lignin from the plant cell walls. This is known as a “lignin-first” approach to breaking down biomass.
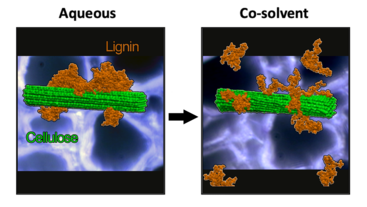
The UC Riverside researchers enlisted the research team at Oak Ridge National Laboratory’s Center of Molecular Biophysics, led by Jeremy Smith, to help construct a 1.5 million atom molecular simulation to reveal how the co-solvent pair consisting of tetrahydrofuran, or THF, and water are particularly effective at altering the interactions between lignin and cellulose, helping to drive multiple key mechanisms responsible for breaking down biomass.
The team discovered that pretreating plant biomass with THF-water caused lignin globules on the cellulose surface to expand and break away from one another and away from the cellulose fibers. The expanded lignin was also more exposed to catalytic fragmentation by dilute acid. As a result, lignin could be more efficiently depolymerized, solubilized, and transported out of the cell wall at milder treatment conditions.
The nearly complete removal of lignin also allowed the remaining cellulose fibers to be more susceptible to enzyme attack. In fact, after mild THF co-solvent treatment, the enzymes added to the remaining cellulose-rich solids achieved complete hydrolysis to glucose sugars.
Collaborating researchers at the University of Central Florida, led by Laurene Tetard, helped to confirm the observations made from the molecular simulations and enzymatic studies by using powerful lasers and nano-infrared imaging to optically track lignin’s rearrangement and removal from the cell wall of micron-thick slices of hardwood.
Finally, Oak Ridge National Laboratory researchers Yunqiao Pu and Arthur Ragauskas showed that lignin extracted from hardwood pretreated with THF co-solvent was significantly depolymerized and contained fewer unwanted reactions than lignin produced from other acidic pretreatment methods.
By putting lignin first, highly functional co-solvents can help to integrate multiple processing steps while allowing both lignin and sugars to be easily recovered as valuable chemical building blocks, making renewable fuel production easier and more cost-effective. The research team hopes that by revealing the synergistic mechanisms of biomass breakdown by co-solvents THF and water, they can inspire others to identify additional multifunctional co-solvent pairs.
The paper, “A multifunctional co-solvent pair reveals molecular principles of biomass deconstruction,” is published in the Journal of the American Chemical Society. In addition to Cai and Patri, authors include Barmak Mostofian; Yunqiao Pu; Nicholas Ciaffone; Mikhael Soliman; Micholas Dean Smith; Rajeev Kumar; Xiaolin Cheng; Charles E. Wyman; Laurene Tetard; Arthur J. Ragauskas; Jeremy C. Smith; and Loukas Petridis.