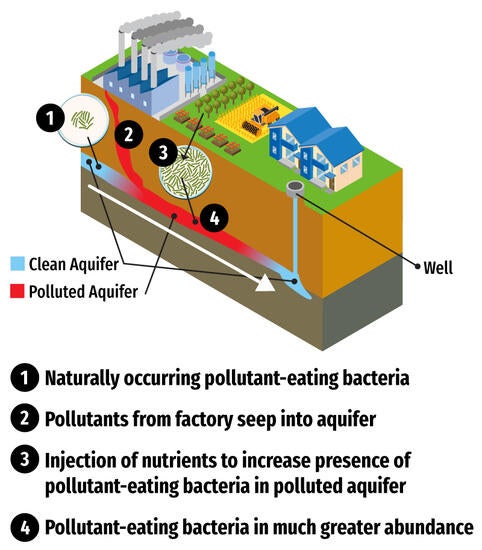
University of California, Riverside, chemical and environmental engineering scientists have identified two species of bacteria found in soil that break down a class of stubborn “forever chemicals,” giving hope for low-cost biological cleanup of industrial pollutants.
These bacteria destroy a subgroup of per- and poly-fluoroalkyl substances, or PFAS, that have one or more chlorine atoms within their chemical structure, Yujie Men, an assistant professor in the Bourns College of Engineering, and her UCR colleagues, reported in the journal Natural Water.
Unhealthful forever chemicals persist in the environment for decades or much longer because of their unusually strong carbon-to-fluorine bonds. Remarkably, the UCR team found that the bacteria cleave the pollutant’s chlorine-carbon bonds, which starts a chain of reactions that destroy the forever chemical structures, rendering them harmless.
“What we discovered is that bacteria can do carbon-chlorine bond cleavage first, generating unstable intermediates,” Men said.
“And then those unstable intermediates undergo spontaneous defluorination, which is the cleavage of the carbon-fluorine bond.”
Chlorinated PFAS are a large group in the forever chemical family of thousands of compounds. They include a variety of non-flammable hydraulic fluids used in industry and compounds used to make chemically stable films that serve as moisture barriers in various industrial, packaging, and electronic applications.
The two bacteria species – Desulfovibrio aminophilus and Sporomusa sphaeroides – identified by Men’s group are naturally occurring and are known to live in the subterranean microbiomes where groundwater may be contaminated with PFAS. For expedited cleanups, an inexpensive nutrient, such as methanol, could be injected into groundwater to promote bacterial growth. This would greatly increase the bacteria’s presence to destroy the pollutants more effectively, Men said. If the bacteria are not already present, the contaminated water could be inoculated with one of the bacterium species.
The title of the paper is “Substantial defluorination of polychlorofluorocarboxylic acids triggered by anaerobic microbial hydrolytic dichlorination.” Men is the corresponding author and Bosen Jin, a UCR chemical and environmental engineering graduate student, is the lead author. Other UCR co-authors are postdoc Jinyu Gao; former postdoc Huaqing Liu; former graduate students Shun Che and Yaochun Yu; and Associate Professor Jinyong Liu.
The preprint version of this paper by Bosen Jin won several awards, including the ACSenvr’s C. Ellen Gonter Award for graduate students in environmental chemistry, the ACSenvr- Society of Environmental Toxicology and Chemistry Exchange Award, and the 2022 Battelle 12th Chlorinated Conference Student Best Paper Award.
The study expands on earlier work by Men, in which she demonstrated that microbes can breakdown a stubborn class of PFAS called fluorinated carboxylic acids.
Microbes have long been used for biological cleanup of oil spills and other industrial pollutants, including the industrial solvent trichloroethylene or TCE, which Men has studied.
But what’s known about using microorganisms to clean up PFAS is still in its infancy, Men said. Her discovery shows great promise because biological treatments, if effective pollutant-eating microbes are available, are generally less costly and more environmentally friendly than chemical treatments. Pollutant-eating microbes can also be injected into difficult-to-reach locations underground.
Men’s latest PFAS study comes as the U.S. Environmental Protection Agency is promulgating new regulations to spur cleanups of PFAS-contaminated groundwater sites throughout the nation because these chemicals have been linked to a host of ill health effects, including cancer, kidney disease, and hormone disruptions.
Starting in the 1940s, PFAS compounds came into widespread use in thousands of consumer products – including grease-resistant paper wrappers and containers, and fire suppressants – because of their ability to form protective coatings that resist heat, water, and lipids.
Identifying the bacteria species that destroy certain forever chemicals was a large step forward, Men said.
“Now, we know who they are,” Men said. “So, we can use the pure cultures to further understand the degradation mechanisms, which enzymes are involved, and whether we can manipulate or modify those to make them better.”
The study was supported by grants from the U.S. Department of Defense’s Strategic Environmental Research and Development Program (ER20-1541) and the National Institute of Environmental Health Sciences (R01 ES032668).